Therapeutic potential of NADH: in neurodegenerative diseases characterizde by mitochondrial dysfunction
-
摘要: 还原型烟酰胺腺嘌呤二核苷酸(nicotinamide adenine dinucleotide,NADH)是能量稳态所必需的氧化还原反应的关键辅酶,参与呼吸链电子传递。NADH和其氧化对应物NAD+被称为氧化还原对,可相互转化,控制多种生物过程,如钙稳态、突触可塑性、抗凋亡和基因表达。NAD+/NADH水平降低与线粒体功能障碍密切相关,而线粒体功能障碍在众多神经退行性病变的级联反应中发挥重要作用,如帕金森病和阿尔茨海默等神经退行性疾病。听神经病(auditory neuropathy,AN)作为神经退行性疾病的一种临床生物学标记,在 OPA1 、 AIFM1 等基因突变为病因的患者中也发现3线粒体功能障碍,但目前尚无有效的治疗方法。越来越多证据表明,内源性给予NAD+或其前体,可通过改善DNA修复损伤、线粒体功能来预防和延缓疾病进展。因此,本综述聚焦于NAD+/NADH的产生代谢途径、生物学功能,探讨了NADH对AN的治疗潜力以及可能的作用机制。
-
关键词:
- 烟酰胺腺嘌呤二核苷酸 /
- 神经退行性疾病 /
- 线粒体功能障碍
Abstract: Nicotinamide adenine dinucleotide(NADH) in its reduced form of is a key coenzyme in redox reactions, essential for maintaining energy homeostasis.NADH and its oxidized counterpart, NAD+, form a redox couple that regulates various biological processes, including calcium homeostasis, synaptic plasticity, anti-apoptosis, and gene expression. The reduction of NAD+/NADH levels is closely linked to mitochondrial dysfunction, which plays a pivotal role in the cascade of various neurodegenerative disorders, including Parkinson's disease and Alzheimer's disease.Auditory neuropathy(AN) is recognized as a clinical biomarker in neurodegenerative disorders. Furthermore, mitochondrial dysfunction has been identified in patients with mutations in genes like OPA1 and AIFM1 . However, effective treatments for these conditions are still lacking. Increasing evidence suggests that administratering NAD+ or its precursors endogenously may potentially prevent and slow disease progression by enhancing DNA repair and improving mitochondrial function. Therefore, this review concentrates on the metabolic pathways of NAD+/NADH production and their biological functions, and delves into the therapeutic potential and mechanisms of NADH in treating AN. -

-
表 1 NADH在临床试验中的应用
疾病 例数 年龄/岁 剂量 疗程 主要结果指标 结果 阿尔茨海默症 1996[45] 17 33~84 10 mg/d 8~12周 简易智力状态检查量;总体衰退量表 该开放标签试验中所有患者经NADH后,都观察其认知功能障碍的改善,但就本试验结果无法得出明确的结论 2000[46] 19 54~91 10 mg/d 12周 简易智力状态检查量;总体衰退量表;阿尔茨海默病评定量表-认知量表 对25例轻度至中度阿尔茨海默氏症、血管性痴呆和额颞型痴呆患者以及目前的胆碱模拟药物治疗患者进行了NADH治疗,并未发现认知改善的迹象 2001[28] 48 50~80 10 mg/d 24周 Mattis痴呆症评定量表总分语言流畅性测试 试验发现NADH能提高AD患者的认知功能 帕金森症 1989[24] 161 / 25 mg/d 10~14 d 尿高香草酸水平;Birkmayer & Neumayer残疾量表 除13例患者外,其余患者失能程度都有不同程度的好转;所有患者尿高香草酸水平升高,即内源性左旋多巴生成增加 1993 885 / / / / 80%患者经NADH治疗后得到改善;治疗前残疾、疾病持续时间和患者年龄与改善程度相关但影响较弱 慢性疲劳综合征 1999[30] 26 20~70 10 mg/d 4周 自研发疲劳调查问卷 试验结果显示30%的患者对NADH反应良好 2004[31] 31 22~54 5或10 mg/d 24个月 疲劳程度问卷以及其他症状问卷;免疫学参数 在试验的前3个月观察到NADH优于常规治疗;多数患者IgG和IgE抗体水平升高 2009[32] 86 18~65 20 mg/d 3个月 疲劳强度;功能状态;情绪;疲劳影响量表;声音质量;疲劳对功能性影响;生活质量 CFS患者服用NADH后焦虑程度以及最大心率下降,但与CFS相关的其他临床表现和生活质量并没有得到改善 2012[47] 58 55.0±1.4 35 mg/d 8周 Piper疲乏量表 服用NADH后患者疲劳改善,8周后继续服用可能进一步缓解 2014[34] 80 49.3±7.1 20 mg/d 8周 疲劳影响量表 补充辅酶Q10和NADH 8周后,CFS患者的疲劳明显改善,FIS总分降低;ATP生成水平提高,线粒体功能改善 2021[33] 207 45.38±7.81/46.79 ±6.48 20 mg/d 12周 疲劳影响量表;匹兹堡睡眠质量指数;36项简明健康调查 NADH和辅酶Q10联用可以有效减少认知疲劳和整体疲劳感知,从而改善ME/CFS中的HRQoL 皮炎 2003[35] 10 21~61 2~3 g 2周 皮肤炎症 6例患者的红斑、水肿和水泡性病变明显减轻,未发现皮肤干燥或脱皮 倒时差 2001[36] 35 35~55 20 mg/d 单次 Kay连续性能测试;分数测试:复杂认知评估组合;自动神经心理学评估指标;沃尔特·里德情绪量表;斯坦福嗜睡量表 服用NADH可改善因时差引起的认知功能紊乱 -
[1] Ransohoff RM. How neuroinflammation contributes to neurodegeneration[J]. Science, 2016, 353(6301): 777-783. doi: 10.1126/science.aag2590
[2] Guerreiro S, Privat AL, Bressac L, et al. CD38 in Neurodegeneration and Neuroinflammation[J]. Cells, 2020, 9(2): 471. doi: 10.3390/cells9020471
[3] Andreone BJ, Larhammar M, Lewcock JW. Cell Death and Neurodegeneration[J]. Cold Spring Harb Perspect Biol, 2020, 12(2): a036434. doi: 10.1101/cshperspect.a036434
[4] Gammon K. Neurodegenerative disease: brain windfall[J]. Nature, 2014, 515(7526): 299-300. doi: 10.1038/nj7526-299a
[5] Verdin E. NAD+ in aging, metabolism, and neurodegeneration[J]. Science, 2015, 350(6265): 1208-1213. doi: 10.1126/science.aac4854
[6] Fang EF, Lautrup S, Hou Y, et al. NAD+ in Aging: Molecular Mechanisms and Translational Implications[J]. Trends Mol Med, 2017, 23(10): 899-916. doi: 10.1016/j.molmed.2017.08.001
[7] Giroud-Gerbetant J, Joffraud M, Giner MP, et al. A reduced form of nicotinamide riboside defines a new path for NAD+ biosynthesis and acts as an orally bioavailable NAD+ precursor[J]. Mol Metab, 2019, 30: 192-202. doi: 10.1016/j.molmet.2019.09.013
[8] Bertoldo MJ, Listijono DR, Ho WJ, et al. NAD+ Repletion Rescues Female Fertility during Reproductive Aging[J]. Cell Rep, 2020, 30(6): 1670-1681. e7. doi: 10.1016/j.celrep.2020.01.058
[9] 刘晓雨. Nadh调控神经突触可塑性基因表达的分子机制[D]. 山东大学, 2020.
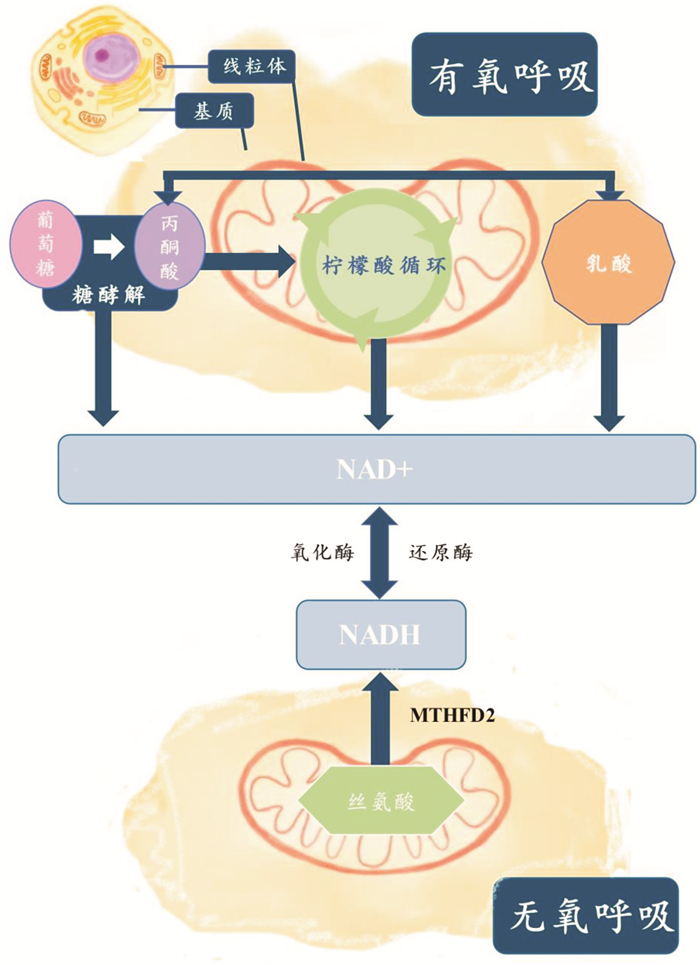
[10] Yang L, Garcia Canaveras JC, Chen Z, et al. Serine Catabolism Feeds NADH when Respiration Is Impaired[J]. Cell Metab, 2020, 31(4): 809-821. e6. doi: 10.1016/j.cmet.2020.02.017
[11] Williamson DH, Lund P, Krebs HA. The redox state of free nicotinamide-adenine dinucleotide in the cytoplasm and mitochondria of rat liver[J]. Biochem J, 1967, 103(2): 514-27. doi: 10.1042/bj1030514
[12] Veech RL, Guynn R, Veloso D. The time-course of the effects of ethanol on the redox and phosphorylation states of rat liver[J]. Biochem J, 1972, 127(2): 387-397. doi: 10.1042/bj1270387
[13] Ying W. NAD+ and NADH in cellular functions and cell death[J]. Front Biosci, 2006, 11: 3129-3148. doi: 10.2741/2038
[14] Ying W. NAD+/NADH and NADP+/NADPH in cellular functions and cell death: regulation and biological consequences[J]. Antioxid Redox Signal, 2008, 10(2): 179-206. doi: 10.1089/ars.2007.1672
[15] Olek RA, Ziolkowski W, Kaczor JJ, et al. Antioxidant activity of NADH and its analogue-an in vitro study[J]. J Biochem Mol Biol, 2004, 37(4): 416-421.
[16] Yamada M, Sakai H. Hidden Antioxidative Functions of Reduced Nicotinamide Adenine Dinucleotide Coexisting with Hemoglobin[J]. ACS Chem Biol, 2017, 12(7): 1820-1829. doi: 10.1021/acschembio.7b00174
[17] Virág L, Szabó C. The therapeutic potential of poly(ADP-ribose)polymerase inhibitors[J]. Pharmacol Rev, 2002, 54(3): 375-429. doi: 10.1124/pr.54.3.375
[18] Zhou Y, Liu L, Tao S, et al. Parthanatos and its associated components: Promising therapeutic targets for cancer[J]. Pharmacol Res, 2021, 163: 105299. doi: 10.1016/j.phrs.2020.105299
[19] Zhu K, Swanson RA, Ying W. NADH can enter into astrocytes and block poly(ADP-ribose)polymerase-1-mediated astrocyte death[J]. Neuroreport, 2005, 16(11): 1209-1212. doi: 10.1097/00001756-200508010-00015
[20] Kharechkina E, Nikiforova A, Kruglov A. NAD(H)Regulates the Permeability Transition Pore in Mitochondria through an External Site[J]. Int J Mol Sci, 2021, 22(16): 8560. doi: 10.3390/ijms22168560
[21] Michán S, Li Y, Chou MM, et al. SIRT1 is essential for normal cognitive function and synaptic plasticity[J]. J Neurosci, 2010, 30(29): 9695-9707. doi: 10.1523/JNEUROSCI.0027-10.2010
[22] Ma XR, Zhu X, Xiao Y, et al. Restoring nuclear entry of Sirtuin 2 in oligodendrocyte progenitor cells promotes remyelination during ageing[J]. Nat Commun, 2022, 13(1): 1225. doi: 10.1038/s41467-022-28844-1
[23] Hou Y, Dan X, Babbar M, et al. Ageing as a risk factor for neurodegenerative disease[J]. Nat Rev Neurol, 2019, 15(10): 565-581. doi: 10.1038/s41582-019-0244-7
[24] Birkmayer GD, Kay GG, Vürre E. [Stabilized NADH(ENADA)improves jet lag-induced cognitive performance deficit][J]. Wien Med Wochenschr, 2002, 152(17-18): 450-454. doi: 10.1046/j.1563-258X.2002.02072.x
[25] Manyam BV, Dhanasekaran M, Hare TA. Neuroprotective effects of the antiparkinson drug Mucuna pruriens[J]. Phytother Res, 2004, 18(9): 706-712. doi: 10.1002/ptr.1514
[26] Hou Y, Lautrup S, Cordonnier S, et al. NAD+ supplementation normalizes key Alzheimer's features and DNA damage responses in a new AD mouse model with introduced DNA repair deficiency[J]. Proc Natl Acad Sci U S A, 2018, 115(8): E1876-E1885.
[27] Hou Y, Wei Y, Lautrup S, et al. NAD+ supplementation reduces neuroinflammation and cell senescence in a transgenic mouse model of Alzheimer's disease via cGAS-STING[J]. Proc Natl Acad Sci USA, 2021, 118(37): e2011226118. doi: 10.1073/pnas.2011226118
[28] V. Demarin1, MD, PhD, et al. ENADA/NADH improves cognitive impairment of Alzheimer patients[G]. First International Conference of Mechanisms of Action of Neutraceuticals, 2001, 22.
[29] Lloret A, Beal MF. PGC-1α, Sirtuins and PARPs in Huntington's Disease and Other Neurodegenerative Conditions: NAD+ to Rule Them All[J]. Neurochem Res, 2019, 44(10): 2423-2434. doi: 10.1007/s11064-019-02809-1
[30] Forsyth LM, Preuss HG, MacDowell AL, et al. Therapeutic effects of oral NADH on the symptoms of patients with chronic fatigue syndrome[J]. Ann Allergy Asthma Immunol, 1999, 82(2): 185-191. doi: 10.1016/S1081-1206(10)62595-1
[31] Santaella ML, Font I, Disdier OM. Comparison of oral nicotinamide adenine dinucleotide(NADH)versus conventional therapy for chronic fatigue syndrome[J]. P R Health Sci J, 2004, 23(2): 89-93.
[32] Alegre J, Rosés JM, Javierre C, et al. [Nicotinamide adenine dinucleotide(NADH)in patients with chronic fatigue syndrome][J]. Rev Clin Esp, 2010, 210(6): 284-288. doi: 10.1016/j.rce.2009.09.015
[33] Castro-Marrero J, Segundo MJ, Lacasa M, et al. Effect of Dietary Coenzyme Q10 Plus NADH Supplementation on Fatigue Perception and Health-Related Quality of Life in Individuals with Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: A Prospective, Randomized, Double-Blind, Placebo-Controlled Trial[J]. Nutrients, 2021, 13(8): 2658. doi: 10.3390/nu13082658
[34] Castro-Marrero J, Cordero MD, Segundo MJ, et al. Does oral coenzyme Q10 plus NADH supplementation improve fatigue and biochemical parameters in chronic fatigue syndrome?[J]. Antioxid Redox Signal, 2015, 22(8): 679-685. doi: 10.1089/ars.2014.6181
[35] Wozniacka A, Sysa-Jedrzejowska A, Adamus J, et al. Topical application of NADH for the treatment of rosacea and contact dermatitis[J]. Clin Exp Dermatol, 2003, 28(1): 61-63. doi: 10.1046/j.1365-2230.2003.01118.x
[36] Birkmayer GD, Kay GG, Vürre E. [Stabilized NADH(ENADA)improves jet lag-induced cognitive performance deficit][J]. Wien Med Wochenschr, 2002, 152(17-18): 450-454. doi: 10.1046/j.1563-258X.2002.02072.x
[37] 王秋菊. Starr A. 听神经病: 从发现到渐入精准[J]. 中华耳鼻咽喉头颈外科杂志, 2018, 53(3): 161-171. https:
[38] 中国听神经病临床诊断与干预多中心研究协作组, 中华耳鼻咽喉头颈外科杂志编辑委员会, 中华医学会耳鼻咽喉头颈外科学分会, 等. 中国听神经病临床实践指南(2022版)[J]. 中华耳鼻咽喉头颈外科杂志, 2022, 57(3): 241-262.
[39] 王秋菊. 儿童听力损失诊治思考[J]. 临床耳鼻咽喉头颈外科杂志, 2023, 37(3): 161-168. https://lceh.whuhzzs.com/article/doi/10.13201/j.issn.2096-7993.2023.03.001
[40] 谢林怡, 熊芬, 兰兰, 等. 儿童听神经病的听力随访研究[J]. 临床耳鼻咽喉头颈外科杂志, 2023, 37(3): 177-180. https://lceh.whuhzzs.com/article/doi/10.13201/j.issn.2096-7993.2023.03.004
[41] Wang H, Bing D, Li J, et al. High Frequency of AIFM1 Variants and Phenotype Progression of Auditory Neuropathy in a Chinese Population[J]. Neural Plast, 2020, 2020: 5625768.
[42] Herrmann JM, Riemer J. Apoptosis inducing factor and mitochondrial NADH dehydrogenases: redox-controlled gear boxes to switch between mitochondrial biogenesis and cell death[J]. Biol Chem, 2021, 402(3): 289-297. doi: 10.1515/hsz-2020-0254
[43] Qiu Y, Wang H, Pan H, et al. AIFM1 variants associated with auditory neuropathy spectrum disorder cause apoptosis due to impaired apoptosis-inducing factor dimerization[J]. J Zhejiang Univ Sci B, 2023, 24(2): 172-184. doi: 10.1631/jzus.B2200081
[44] Ravera S, Bartolucci M, Cuccarolo P, et al. Oxidative stress in myelin sheath: The other face of the extramitochondrial oxidative phosphorylation ability[J]. Free Radic Res, 2015, 49(9): 1156-1164. doi: 10.3109/10715762.2015.1050962
[45] Birkmayer JG. Coenzyme nicotinamide adenine dinucleotide: new therapeutic approach for improving dementia of the Alzheimer type[J]. Ann Clin Lab Sci, 1996, 26(1): 1-9.
[46] Rainer M, Kraxberger E, Haushofer M, et al. No evidence for cognitive improvement from oral nicotinamide adenine dinucleotide(NADH)in dementia[J]. J Neural Transm(Vienna), 2000, 107(12): 1475-1481. doi: 10.1007/s007020070011
[47] Nicolson GL, Settineri R, Ellithorpe R. Lipid replacement therapy with a glycophospholipid formulation with nadh and coq10 significantly reduces fatigue in intractable chronic fatiguing illnesses and chronic lyme disease patients[J]. Int J Clin Med, 2012, 3(3): 8.
-




 下载:
下载: