Analysis of efficacy and prognosis of neoadjuvant chemotherapy and (or) surgery plus radiotherapy for hypopharyngeal squamous cell carcinoma
-
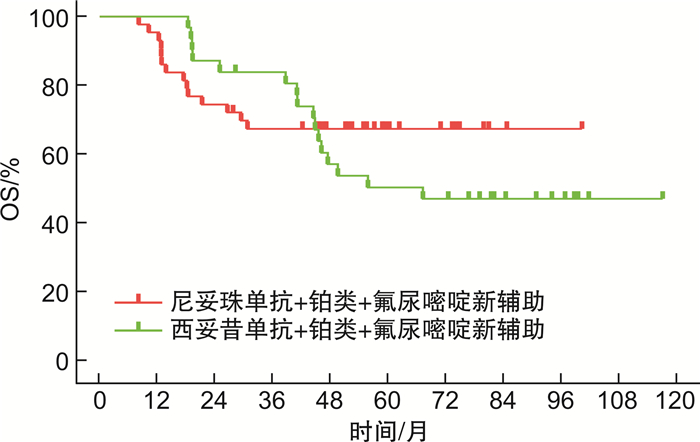
摘要: 目的 分析影响下咽鳞状细胞癌(hypopharyngeal squamous cell carcinoma,HPSCC)患者预后的危险因素;比较手术+术后根治性放疗(SR)治疗、尼妥珠单抗/西妥昔单抗联合铂类+氟尿嘧啶新辅助化疗+SR治疗对患者的总生存期(overall surviva,OS)、术后进食情况、气管套管拔除率以及2种新辅助化疗的肿瘤缓解情况。方法 回顾性分析2012至2019年上海交通大学医学院附属第一人民医院就诊且既往尚未接受治疗,入院后行SR或新辅助化疗+SR治疗的HPSCC患者资料。分析预后影响因素,进行2种新辅助化疗方案对行SR患者的生存分析。结果 纳入108例患者,单因素分析结果显示性别(P=0.850)对接受SR的HPSCC患者的生存率无明显相关性,而年龄、吸烟史、饮酒史、血小板与淋巴细胞计数比(platelet to lymphocyte ratio,PLR)、中性粒细胞与淋巴细胞计数比(neutrophil to lymphocyte ratio,NLR)、T分期、N分期、尼妥珠单抗/西妥昔单抗+铂类+氟尿嘧啶新辅助化疗、病理分级与预后相关(P < 0.05)。多因素分析显示其中吸烟史、病理分级、尼妥珠单抗/西妥昔单抗+铂类+氟尿嘧啶新辅助化疗是影响HPSCC预后的独立危险因素(P < 0.05)。经过新辅助化疗的患者OS长于仅SR治疗的患者(P < 0.001)。2种新辅助化疗肿瘤缓解情况和OS比较, 差异无统计学意义(P>0.05),且3种治疗方案的患者经口进食情况和气管套管拔管情况比较差异无统计学意义(P>0.05)。结论 单因素分析显示肿瘤发病年龄、吸烟史、饮酒史、NLR、PLR、T分期、N分期、是否接受新辅助化疗和病理分级等与SR治疗的HPSCC患者预后相关;且多因素分析显示吸烟史、病理分级及新辅助化疗是影响预后的独立危险因素,联合尼妥珠单抗/西妥昔单抗的新辅助化疗可延长患者OS,为HPSCC的治疗提供了一定的依据和参考。Abstract: Objective To analyze the risk factors that affect the prognosis of patients with hypopharyngeal squamous cell carcinoma(HPSCC) and to compare the efficacy of surgical resection followed by adjuvant radiotherapy(SR) with that of neoadjuvant therapy consisting of platinum-based chemotherapy and fluorouracil combined with either cetuximab or nimotuzumab, followed by SR. The study also aimed to evaluate the overall survival(OS) of patients, their postoperative eating function, tracheostomy decannulation rate, and tumor response to the two neoadjuvant chemotherapies.Methods A retrospective analysis was performed on the medical records of HPSCC patients who received SR or neoadjuvant therapy followed by SR treatment at the Shanghai General Hospital from 2012 to 2019 and had not undergone any prior treatment. The prognostic factors were analyzed, and the survival analysis of patients who underwent SR treatment with two neoadjuvant chemotherapy regimens was performed.Results A total of 108 patients were included in the study. The results of the univariate analysis showed that gender(P=0.850) had no significant correlation with the survival rate of HPSCC patients who underwent SR. However, age, smoking history, alcohol consumption history, platelet-to-lymphocyte ratio(PLR), neutrophil-to-lymphocyte ratio(NLR), T stage, N stage, neoadjuvant therapy with either cetuximab or nimotuzumab combined with platinum-based chemotherapy and fluorouracil, and histological grade were significantly associated with prognosis(P < 0.05). The multivariate analysis revealed that smoking history, histological grade, and neoadjuvant therapy with either cetuximab or nimotuzumab combined with platinum-based chemotherapy and fluorouracil were independent risk factors affecting the prognosis of HPSCC(P < 0.05). Patients who received neoadjuvant therapy had longer OS than those who underwent SR only(P < 0.001). There was no significant difference in tumor response to the two neoadjuvant therapies and in OS(P>0.05), and there was no significant difference in the rate of oral feeding and tracheostomy decannulation among the three treatment groups(P>0.05).Conclusion Univariate analysis showed that age at tumor onset, smoking history, alcohol consumption history, NLR, PLR, T stage, N stage, whether receiving neoadjuvant chemotherapy, and pathological grade were associated with the prognosis of HPSCC patients receiving SR treatment. Multivariate analysis showed that smoking history, pathological grade, and neoadjuvant chemotherapy were independent risk factors affecting the prognosis. Neoadjuvant chemotherapy with cetuximab or nimotuzumab can prolong the OS of patients, providing a certain basis and reference for the treatment of HPSCC.
-

-
表 1 与HPSCC预后相关的单因素Cox回归分析(n=108)
指标 P HR 95%CI 性别 0.850 1.145 0.279~4.698 发病年龄 0.007 0.450 0.252~0.805 吸烟史 < 0.001 5.778 2.957~11.293 饮酒史 < 0.001 3.776 2.017~7.070 NLR 0.002 1.332 1.107~1.604 PLR 0.002 1.007 1.003~1.011 T分期 0.023 1.832 1.088~3.085 N分期 0.006 2.120 1.246~3.605 是否接受新辅助化疗 < 0.001 是否接受尼妥珠单抗+铂类+氟尿嘧啶新辅助化疗 0.003 0.388 0.208~0.722 是否接受西妥昔单抗+铂类+氟尿嘧啶新辅助化疗 < 0.001 0.288 0.151~0.552 病理分级 0.008 0.434 0.235~0.801 表 2 与HPSCC预后相关多因素Cox回归分析
指标 回归系数 标准误 HR 95%CI P 是否接受新辅助化疗 0.002 是否接受尼妥珠单抗+铂类+氟尿嘧啶新辅助化疗 -0.896 0.342 0.408 0.209~0.798 0.009 是否接受西妥昔单抗+铂类+氟尿嘧啶新辅助化疗 -1.083 0.344 0.339 0.173~0.664 0.002 发病年龄 -0.581 0.315 0.559 0.301~1.037 0.065 吸烟史 1.141 0.484 3.129 1.211~8.082 0.018 饮酒史 0.766 0.466 2.150 0.862~5.362 0.101 NLR 0.236 0.140 1.266 0.963~1.664 0.091 PLR 0.002 0.003 1.002 0.996~1.008 0.475 T分期 0.432 0.321 1.540 0.821~2.891 0.179 N分期 0.354 0.284 1.425 0.816~2.486 0.213 病理分级 -0.703 0.334 0.495 0.257~0.952 0.035 表 3 接受新辅助化疗组与未接受新辅助化疗组基线情况比较
例(%) 变量 接受新辅助化疗组
(n=74)未接受新辅助化疗组
(n=34)P 年龄 ≤60岁 41(55.4) 24(70.6) 0.134 >60岁 33(44.6) 10(29.4) 性别 男 71(95.9) 33(97.1) 1.000 女 3(4.1) 1(2.9) T分期 T1+T2期 45(60.8) 17(50.0) 0.291 T3+T4期 29(39.2) 17(50.0) N分期 N0+N1期 39(52.7) 16(47.1) 0.586 N2+N3期 35(47.3) 18(52.9) 病理分级 低分化 11(14.9) 7(20.6) 0.643 中分化+高分化 63(85.1) 27(79.4) 吸烟史 无 36(48.6) 12(35.3) 0.195 有 38(51.4) 22(64.7) 饮酒史 无 32(43.2) 16(47.1) 0.711 有 42(56.8) 18(52.9) 表 4 2种新辅助化疗方案的肿瘤缓解情况比较
例 肿瘤缓解情况 接受尼妥珠单抗+铂类+氟尿嘧啶新辅助化疗2个周期(n=31) 接受西妥昔单抗+铂类+氟尿嘧啶新辅助化疗2个周期(n=43) PR 16 26 SD 15 17 -
[1] Hall SF, Groome PA, Irish J, et al. The natural history of patients with squamous cell carcinoma of the hypopharynx[J]. Laryngoscope, 2008, 118(8): 1362-1371. doi: 10.1097/MLG.0b013e318173dc4a
[2] 王欢, 高骞, 于亚峰. 长链非编码RNA在下咽鳞状细胞癌发病机制中的作用[J]. 国际耳鼻咽喉头颈外科杂志, 2022, 46(1): 36-40.
[3] Li Y, Ou X, Hu C. Prevalence and prognostic impact of synchronous distant metastases in patients with hypopharynx squamous cell carcinomas: a SEER-based study[J]. J Cancer, 2019, 10(3): 620-626. doi: 10.7150/jca.28554
[4] Eckel HE, Bradley PJ. Future Perspectives in Hypopharyngeal Cancer Care[J]. Adv Otorhinolaryngol, 2019, 83: 167-175.
[5] Global Burden of Disease Cancer Collaboration; Fitzmaurice C, Abate D, et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-Years for 29 Cancer Groups, 1990 to 2017: A Systematic Analysis for the Global Burden of Disease Study[J]. JAMA Oncol, 2019, 5(12): 1749-1768. doi: 10.1001/jamaoncol.2019.2996
[6] Li Y, Ou X, Hu C. Prevalence and prognostic impact of synchronous distant metastases in patients with hypopharynx squamous cell carcinomas: a SEER-based study[J]. J Cancer, 2019, 10(3): 620-626. doi: 10.7150/jca.28554
[7] Habib A. Management of advanced hypopharyngeal carcinoma: systematic review of survival following surgical and non-surgical treatments[J]. J Laryngol Otol, 2018, 132(5): 385-400. doi: 10.1017/S0022215118000555
[8] Haerle SK, Schmid DT, Ahmad N, et al. The value of F-18-FDG PET/CT for the detection of distant metastases in high-risk patients with head and neck squamous cell carcinoma[J]. Oral Oncology, 2011, 47(7): 653-659. doi: 10.1016/j.oraloncology.2011.05.011
[9] Iwatsubo T, Ishihara R, Morishima T, et al. Impact of age at diagnosis of head and neck cancer on incidence of metachronous cancer[J]. BMC Cancer, 2019, 19(1): 3. doi: 10.1186/s12885-018-5231-7
[10] Takes RP, Rinaldo A, Silver CE, et al. Distant metastases from head and neck squamous cell carcinoma. Part Ⅰ. Basic aspects[J]. Oral Oncology, 2012, 48(9): 775-779. doi: 10.1016/j.oraloncology.2012.03.013
[11] Thakur K, Singh CA, Thakar A, et al. Prevalence of Synchronous ESCN in Head and Neck Cancer: A Single-Institution Perspective[J]. Laryngoscope, 2021, 131(3): E807-E14.
[12] Tseng CM, Wang HH, Lee CT, et al. A nationwide population-based study to access the risk of metachronous esophageal cancers in head and neck cancer survivors[J]. Sci Rep, 2020, 10(1): 884. doi: 10.1038/s41598-020-57630-6
[13] Sewnaik A, Baatenburg DE, Jong RJ. Sequelae and Complications of Treatment for Hypopharyngeal Cancer: Minimising the Risks[J]. Adv Otorhinolaryngol, 2019, 83: 109-117.
[14] Bozec A, Poissonnet G, Dassonville O, et al. Current Therapeutic Strategies for Patients with Hypopharyngeal Carcinoma: Oncologic and Functional Outcomes[J]. J Clin Med, 2023, 12(3): 1237. doi: 10.3390/jcm12031237
[15] Mattei P, Thamphya B, Chamorey E, et al. Therapeutic strategies, oncologic and swallowing outcomes and their predictive factors in patients with locally advanced hypopharyngeal cancer[J]. Eur Arch Oto-Rhino-L, 2022, 279(7): 3629-3637. doi: 10.1007/s00405-021-07196-4
[16] 杨伟光, 曹华, 王刘中, 等. 基于SEER数据库的下咽癌预后模型的建立与分析[J]. 肿瘤基础与临床, 2022, 35(3): 231-236. https://www.cnki.com.cn/Article/CJFDTOTAL-HLZL202203012.htm
[17] 雷倩, 孙传政, 陈冉, 等. 老年头颈部鳞状细胞癌患者的治疗耐受性及预后情况分析[J]. 癌症进展, 2021, 19(17): 1758-1762. https://www.cnki.com.cn/Article/CJFDTOTAL-AZJZ202117009.htm
[18] Guo W, Lu X, Liu Q, et al. Prognostic value of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio for breast cancer patients: An updated meta-analysis of 17079 individuals[J]. Cancer Med, 2019, 8(9): 4135-4148. doi: 10.1002/cam4.2281
[19] Ohe Y, Fushida S, Yamaguchi T, et al. Peripheral Blood Platelet-Lymphocyte Ratio Is Good Predictor of Chemosensitivity and Prognosis in Gastric Cancer Patients[J]. Cancer Management and Research, 2020, 12: 1303-1311. doi: 10.2147/CMAR.S241069
[20] Hu B, Yang XR, Xu Y, et al. Systemic Immune-Inflammation Index Predicts Prognosis of Patients after Curative Resection for Hepatocellular Carcinoma[J]. Clinical Cancer Research, 2014, 20(23): 6212-6222. doi: 10.1158/1078-0432.CCR-14-0442
[21] De Giorgi U, Mego M, Scarpi E, et al. Association between circulating tumor cells and peripheral blood monocytes in metastatic breast cancer[J]. Ther Adv Med Oncol, 2019, 11.
[22] Anderson NM, Simon MC. The tumor microenvironment[J]. Current Biology, 2020, 30(16): R921-R5. doi: 10.1016/j.cub.2020.06.081
[23] Fukuda N, Wang X, Ohmoto A, et al. Sequential Analysis of Neutrophil-to-lymphocyte Ratio for Differentiated Thyroid Cancer Patients Treated With Lenvatinib[J]. In Vivo, 2020, 34(2): 709-714. doi: 10.21873/invivo.11828
[24] Hu X, Tian T, Zhang X, et al. Neutrophil-to-lymphocyte and hypopharyngeal cancer prognosis: System review and meta-analysis[J]. Head Neck, 2023, 45(2): 492-502. doi: 10.1002/hed.27246
[25] Kuo C, Hsueh WT, Wu YH, et al. The Role of Pretreatment Serum Neutrophil-to-Lymphocyte Ratio in Hypopharyngeal Cancer Treated with Definitive Chemoradiotherapy: A Pilot Study[J]. Sci Rep, 2019, 9(1): 1618. doi: 10.1038/s41598-018-38282-z
[26] Wang J, Wang S, Song X, et al. The prognostic value of systemic and local inflammation in patients with laryngeal squamous cell carcinoma[J]. Onco Targets Ther, 2016, 9: 7177-7185. doi: 10.2147/OTT.S113307
[27] Menter DG, Kopetz S, Hawk E, et al. Platelet "first responders" in wound response, cancer, and metastasis[J]. Cancer Metast Rev, 2017, 36(2): 199-213. doi: 10.1007/s10555-017-9682-0
[28] Chen H, Song SH, Li A, et al. Presurgical platelet-lymphocyte ratio for prognosis in advanced hypopharyngeal squamous cell carcinoma in individuals undergoing radical resection[J]. Acta Oto-Laryngol, 2021, 141(5): 537-543. doi: 10.1080/00016489.2021.1891456
[29] Wan M, Zhao D, Liu WX, et al. Pretherapy platelet-to-lymphocyte ratio as a prognostic parameter for locally advanced hypopharyngeal cancer patients treated with radiotherapy combined with chemotherapy[J]. Eur Arch Oto-Rhino-L, 2022, 279(12): 5859-5868. doi: 10.1007/s00405-022-07495-4
[30] 何雨蓉, 钟琦, 王元, 等. 诱导化疗及分子靶向治疗在局部晚期喉癌和下咽癌治疗中的应用[J]. 国际耳鼻咽喉头颈外科杂志, 2022, 46(1): 32-35.
[31] Eisenberger M, Hornedo J, Silva H, et al. Carboplatin(NSC-241-240): an active platinum analog for the treatment of squamous-cell carcinoma of the head and neck[J]. J Clin Oncol, 1986, 4(10): 1506-1509. doi: 10.1200/JCO.1986.4.10.1506
[32] Wei XF, Srivastava A, Lin P, et al. Neoadjuvant chemotherapy as a comprehensive treatment in patients with laryngeal and hypopharyngeal carcinoma[J]. Acta Oto-Laryngol, 2020, 140(7): 603-609. doi: 10.1080/00016489.2020.1738548
[33] Solimeno LS, Park YM, Lim JY, et al. Treatment outcomes of neoadjuvant chemotherapy and transoral robotic surgery in locoregionally advanced laryngopharyngeal carcinoma[J]. Head Neck, 2021, 43(11): 3429-3436. doi: 10.1002/hed.26838
[34] Echanique KA, Evans LK, Han AY, et al. Cancer of the Larynx and Hypopharynx[J]. Hematol Oncol Clin North Am, 2021, 35(5): 933-947. doi: 10.1016/j.hoc.2021.05.005
[35] Janoray G, Pointreau Y, Alfonsi M, et al. Induction chemotherapy followed by cisplatin or cetuximab concomitant to radiotherapy for laryngeal/hypopharyngeal cancer: Long-term results of the TREMPLIN randomised GORTEC trial[J]. Eur J Cancer, 2020, 133: 86-93. doi: 10.1016/j.ejca.2020.04.009
[36] Dietz A, Wichmann G, Kuhnt T, et al. Induction chemotherapy(IC)followed by radiotherapy(RT)versus cetuximab plus IC and RT in advanced laryngeal/hypopharyngeal cancer resectable only by total laryngectomy-final results of the larynx organ preservation trial DeLOS-Ⅱ[J]. Ann Oncol, 2018, 29(10): 2105-2114. doi: 10.1093/annonc/mdy332
[37] 董频, 房居高, 高军茂, 等. 下咽癌外科手术及综合治疗专家共识[J]. 中华耳鼻咽喉头颈外科杂志, 2017, 52(1): 16-24.
[38] Tian X, Xuan Y, Wu R, et al. Nimotuzumab Combined with Induction Chemotherapy and Concurrent Chemoradiotherapy in Unresectable Locally Advanced Hypopharyngeal Carcinoma: A Single Institution Experience in China[J]. Cancer Manag Res, 2020, 12: 3323-3329. doi: 10.2147/CMAR.S248392
[39] Perez R, Moreno E, Garrido G, et al. EGFR-Targeting as a Biological Therapy: Understanding Nimotuzumab's Clinical Effects[J]. Cancers(Basel), 2011, 3(2): 2014-2031.
-





 下载:
下载: