-
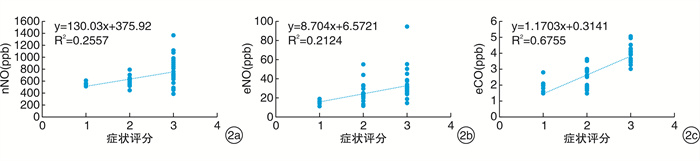
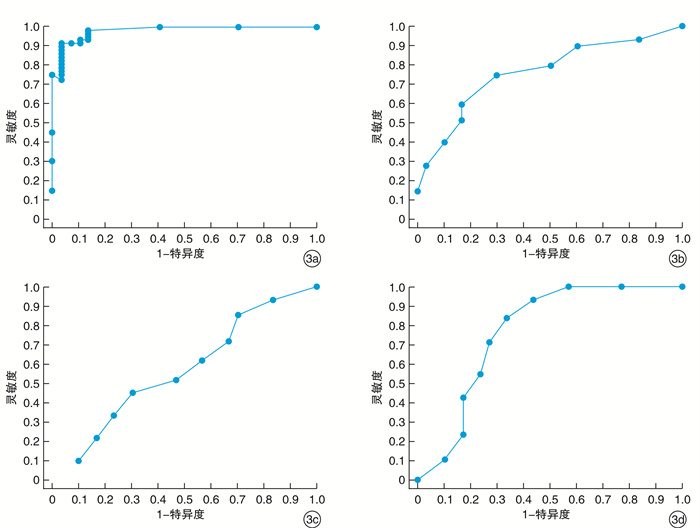
摘要: 目的 通过检测变应性鼻炎(AR)患者鼻呼出一氧化氮(nNO)、口呼出一氧化氮(eNO)及口呼出一氧化碳(eCO)水平, 探讨呼出NO及CO与AR的相关性。方法 选择AR患者60例(AR组), 根据症状评分再分为轻、中、重度亚组, 健康志愿者30人(对照组)。检测AR组、各AR亚组和对照组nNO、eNO及eCO水平。结果 AR组nNO、eNO及eCO水平较对照组升高(P < 0.05), nNO、eNO及eCO水平与症状评分均呈正相关(P < 0.05)。轻、中、重度AR亚组间相比较, nNO与eNO水平差异均有统计学意义, 重度>中度>轻度(P < 0.05), 但轻度AR亚组与对照组eNO水平无明显差异(P>0.05);eCO的结果显示重度AR亚组eCO较中、轻度AR亚组高(P < 0.05), 轻、中度AR组eNO与对照组之间两两比较差异均无统计学意义(P>0.05)。将nNO、eNO及eCO水平作为评估AR病情程度的指标, 绘制受试者ROC曲线, AUC分别为0.978, 0.786和0.577, eCO水平作为评估重度AR的指标, AUC为0.728, 表明nNO对AR病情程度的评估具有较高的准确性, eNO对AR、eCO对重度AR的病情评估有一定的准确性。结论 nNO、eNO及eCO水平检测可作为一种客观的AR病情评估方法。Abstract: Objective In this study, the level of nasal nitric oxide(nNO), exhaled nitric oxide(eNO), and exhaled carbon monoxide(eCO) in patients with allergic rhinitis(AR) were tested, to explore the correlation between nNO, eNO, eCO and AR.Method A total of 60 AR patientswere enrolled as the allergy group and then divided into mild, medium and severe subgroups according to symptom scores. 30 healthy volunteers were recruited as control group. The levels of nNO, eNO and eCO were detected in AR group, AR subgroups and control group.Result The levels of nNO, eNO and eCO in AR group were higher than those in control group(P < 0.05). nNO, eNO and eCO levels were positively correlated with symptom scores(P < 0.05).Pairwise comparison was used between mild, moderate and severe subgroups. The difference of both nNO and eNO levels between mild, moderate and severe AR subgroups was statistically significant, severe>and moderate>were mild(P < 0.05), but there was no significant difference in eNO levels between the mild AR subgroup and the control group(P>0.05). Level of eCO in severe AR subgroup was higher than those in moderate and mild AR subgroups(P < 0.05), but there was no significant difference between mild, moderate AR subgroups and control group(P>0.05). nNO, eNO and eCO levels were used as indicators to evaluate the severity of AR, and the receiver operating characteristic(ROC) curves were plotted. The area under curve(AUC) was 0.978, 0.786 and 0.577, respectively. Taking eCO level as the indicator for disease assessment of severe AR, the AUC was 0.728. It showed that nNO had a high accuracy in evaluating the severity of AR, eNO had a certain accuracy in evaluating the severity of AR, and eCO had a certain accuracy in the assessment of severe AR.Conclusion The detection of nNO, eNO and eCO levels can be used as an objective assessment method of the severity of AR.
-
Key words:
- allergic rhinitis /
- nasal nitric oxide /
- exhaled nitric oxide /
- exhaled carbon monox
-

-
表 1 AR组和对照组基线水平比较
x±s 组别 例数 年龄 性别 身高/m 体重/kg 男 女 AR组 60 25.0±6.3 24 36 174.3±9.2 68.2±11.0 对照组 30 27.0±8.4 13 17 176.2±7.5 67.1±8.8 t 0.834 0.512 0.576 0.422 P 0.365 0.705 0.665 0.831 表 2 AR组与对照组nNO、eNO及eCO的比较
x±s,ppb 组别 例数 nNO eNO eCO AR组 60 685.83±182.41 27.31±13.40 3.10±1.01 对照组 30 301.83±134.68 16.67±6.40 2.64±1.09 t 10.107 4.079 2.372 P 0.001 0.006 0.049 表 3 各AR亚组及对照组nNO、eNO、eCO水平ppb
组别 例数 nNO eNO eCO 重度组 31 776.55±210.42 32.58±14.51 3.93±0.52 中度组 21 664.10±98.85 24.29±10.29 2.33±0.58 轻组度 8 519.50±46.04 14.88±2.59 1.90±0.43 对照组 30 301.83±136.98 16.67±6.40 2.64±1.09 -
[1] 韩德民, 张罗, 黄丹, 等. 我国11个城市变应性鼻炎自报患病率调查[J]. 中华耳鼻咽喉头颈外科杂志, 2007, 42(5): 378-384. doi: 10.3760/j.issn:1673-0860.2007.05.015
[2] Fokkens WJ, Lund VJ, Mullol J, et al. European Position Paper on Rhinosinusitis and Nasal Polyps 2012[J]. Rhinol Suppl, 2012, 23: 1-298.
[3] Kawamoto H, Takeno S, Yajin K. Increased expression of inducible nitric oxide synthase in nasal epithelial cells in patients with allergic rhinitis[J]. Laryngoscope, 1999, 109(12): 2015-2020. doi: 10.1097/00005537-199912000-00023
[4] Arnal JF, Didier A, Rami J, et al. Nasal nitric oxide is increased in allergic rhinitis[J]. Clin Exp Allergy, 1997, 27(4): 358-362. doi: 10.1111/j.1365-2222.1997.tb00719.x
[5] Wang X, Du K, She W, et al. Recent advances in the diagnosis of allergic rhinitis[J]. Expert Rev Clin Immunol, 2018, 14(11): 957-964. doi: 10.1080/1744666X.2018.1530113
[6] Wheatley LM, Togias A. Clinical practice. Allergic rhinitis[J]. N Engl J Med, 2015, 372(5): 456-463. doi: 10.1056/NEJMcp1412282
[7] Seidman MD, Gurgel RK, Lin SY, et al. Clinical practice guideline: allergic rhinitis executive summary[J]. Otolaryngol Head Neck Surg, 2015, 152(2): 197-206. doi: 10.1177/0194599814562166
[8] Pfaar O, Demoly P, Gerth van Wijk R, et al. Recommendations for the standardization of clinical outcomes used in allergen immunotherapy trials for allergic rhinoconjunctivitis: an EAACI Position Paper[J]. Allergy, 2014, 69(7): 854-867. doi: 10.1111/all.12383
[9] American Thoracic Society, European Respiratory Society. ATS/ERS recommendations for standardized procedures for the online and offline measurement of exhaled lower respiratory nitric oxide and nasal nitric oxide, 2005[J]. Am J Respir Crit Care Med, 2005, 171(8): 912-930. doi: 10.1164/rccm.200406-710ST
[10] Lee KJ, Cho SH, Lee SH, et al. Nasal and exhaled nitric oxide in allergic rhinitis[J]. Clin Exp Otorhinolaryngol, 2012, 5(4): 228-233. doi: 10.3342/ceo.2012.5.4.228
[11] Olin AC, Rosengren A, Thelle DS, et al. Increased fraction of exhaled nitric oxide predicts new-onset wheeze in a general population[J]. Am J Respir Crit Care Med, 2010, 181(4): 324-327. doi: 10.1164/rccm.200907-1079OC
[12] Takeno S, Okabayashi Y, Kohno T, et al. The role of nasal fractional exhaled nitric oxide as an objective parameter independent of nasal airflow resistance in the diagnosis of allergic rhinitis[J]. Auris Nasus Larynx, 2017, 44(4): 435-441. doi: 10.1016/j.anl.2016.09.007
[13] Mancini AL, Regina da Silva Carvalho S, Valente de Crasto Mdo C, et al. Echocardiograph alterations in asthma patients[J]. Rev Port Pneumol, 2008, 14(3): 363-377. doi: 10.1016/S0873-2159(15)30244-0
[14] Monma M, Yamaya M, Sekizawa K, et al. Increased carbon monoxide in exhaled air of patients with seasonal allergic rhinitis[J]. Clin Exp Allergy, 1999, 29(11): 1537-1541. doi: 10.1046/j.1365-2222.1999.00684.x
[15] Yu SQ, Zhang RX, Chen YJ, et al. A meta-analysis of the association of exhaled carbon monoxide on asthma and allergic rhinitis[J]. Clin Rev Allergy Immunol, 2011, 41(1): 67-75. doi: 10.1007/s12016-009-8195-1
-

| 引用本文: | 王扬, 秦马力, 金玲, 等. 呼出一氧化氮及一氧化碳与变应性鼻炎的相关性研究[J]. 临床耳鼻咽喉头颈外科杂志, 2020, 34(11): 1014-1018. doi: 10.13201/j.issn.2096-7993.2020.11.013 |
| Citation: | WANG Yang, QIN Mali, JIN Ling, et al. Correlation between exhaled nitric oxide and carbon monoxide and allergic rhinitis[J]. J Clin Otorhinolaryngol Head Neck Surg, 2020, 34(11): 1014-1018. doi: 10.13201/j.issn.2096-7993.2020.11.013 |
- Figure 1.
- Figure 2.
- Figure 3.




 下载:
下载: